Medical Device Certification Courses
Medical Device Certification Courses - We select talented and driven individuals for admissions into the prepmd cardiac medical device specialist training program. Accurate classification of medical device risk levels is essential for regulatory oversight and clinical safety. The dual certificate program in medical devices and pharmaceuticals. The cbet certification validates expertise in testing, calibrating, and maintaining medical. This training will similarities and differences in. Meet the challenges of expanding regulations. Free online courses for state, local, and tribal regulatory. Increase your understanding of specific topics shaping the medtech sector through our wide range of highly specialized courses. Learn fda cgmp regulations, quality system requirements, risk management, and compliance best. Iso 14971 risk management online certified course designed for medical device professionals. Overview of our pharmaceutical, medical device, and biotechnology certificate programs categorized by industry. Online courses for industry on safety and effectiveness of medical devices and exposure to radiation from medical devices. Your company’s effectiveness depends on your employees’ skills and knowledge. Course tutors selected from the following: Prepmd students learn the clinical, technical, and business. Learn about the medical device industry and regulations at your own. Accurate classification of medical device risk levels is essential for regulatory oversight and clinical safety. Transfer friendlycourses designed for yourigorous curriculumlearn valuable skills Our training covers risk management, pharmaceutical,. Analyze the regulatory requirements for class i, ii, and iii devices. 8 june 2025 the world health organization (who) is seeking experts to serve as members of the strategic and technical advisory group on. Online courses for industry on safety and effectiveness of medical devices and exposure to radiation from medical devices. How to register, etc., please contact. Earn a recognized certificate and cpd/ceu credits upon completion. Transfer friendlycourses designed for. Earn a recognized certificate and cpd/ceu credits upon completion. Free online courses for state, local, and tribal regulatory. Understand the fda classification system for medical devices. Our training covers risk management, pharmaceutical,. Accurate classification of medical device risk levels is essential for regulatory oversight and clinical safety. Medical device reprocessing acknowledged by ipac canada type: Accurate classification of medical device risk levels is essential for regulatory oversight and clinical safety. Course tutors selected from the following: The dual certificate program in medical devices and pharmaceuticals. Build a solid body of regulatory knowledge and advance your career with a raps online university certificate. How to register, etc., please contact. Understand the fda classification system for medical devices. Online courses for industry on safety and effectiveness of medical devices and exposure to radiation from medical devices. Overview of our pharmaceutical, medical device, and biotechnology certificate programs categorized by industry. Learn industry standards, requirements and best practices by our team of experts from the medical. Prepmd students learn the clinical, technical, and business. Our training covers risk management, pharmaceutical,. Learn industry standards, requirements and best practices by our team of experts from the medical device industry. We select talented and driven individuals for admissions into the prepmd cardiac medical device specialist training program. Medical device reprocessing acknowledged by ipac canada type: Document your dedication to medical devices by earning a professional certification from biopharma institute. Increase your understanding of specific topics shaping the medtech sector through our wide range of highly specialized courses. Overview of our pharmaceutical, medical device, and biotechnology certificate programs categorized by industry. Earn a recognized certificate and cpd/ceu credits upon completion. We select talented and driven individuals. The dual certificate program in medical devices and pharmaceuticals. Welcome to the medical device single audit program (mdsap) compliance course. Medical device reprocessing acknowledged by ipac canada type: This training will similarities and differences in. Learn fda cgmp regulations, quality system requirements, risk management, and compliance best. Free online courses for state, local, and tribal regulatory. Medical device reprocessing acknowledged by ipac canada type: Build a solid body of regulatory knowledge and advance your career with a raps online university certificate. Medical sales college is a licensed private, postsecondary institution, training the next generation of medical sales reps. Document your dedication to medical devices by earning a. Medical sales college is a licensed private, postsecondary institution, training the next generation of medical sales reps. The dual certificate program in medical devices and pharmaceuticals. Our training covers risk management, pharmaceutical,. Medical device reprocessing acknowledged by ipac canada type: The cbet certification validates expertise in testing, calibrating, and maintaining medical. Free online courses for state, local, and tribal regulatory. Learn about the medical device industry and regulations at your own. Prepmd students learn the clinical, technical, and business. Course tutors selected from the following: The cbet certification validates expertise in testing, calibrating, and maintaining medical. Analyze the regulatory requirements for class i, ii, and iii devices. Our training covers risk management, pharmaceutical,. The cbet certification validates expertise in testing, calibrating, and maintaining medical. Welcome to the medical device single audit program (mdsap) compliance course. Overview of our pharmaceutical, medical device, and biotechnology certificate programs categorized by industry. How to register, etc., please contact. Build a solid body of regulatory knowledge and advance your career with a raps online university certificate. 8 june 2025 the world health organization (who) is seeking experts to serve as members of the strategic and technical advisory group on. Your company’s effectiveness depends on your employees’ skills and knowledge. Learn about the medical device industry and regulations at your own. Transform you career with coursera's online medical device courses. Earn a recognized certificate and cpd/ceu credits upon completion. Free online courses for state, local, and tribal regulatory. Medical device reprocessing acknowledged by ipac canada type: Prepmd students learn the clinical, technical, and business. Transfer friendlycourses designed for yourigorous curriculumlearn valuable skillsWorkforce Learning Solutions Hurix Digital
Medical Device Training Courses Medical Device HQ
PPT ISO 13485 QMS Medical Device Certification PowerPoint
Certification Of Medical Devices
Achieve More with Medical Device Training and Certifications Medical
Medical Arts Pharmacy Fayetteville Arkansas Medical Device Training
Medical Device Medical Representative Training Royed Training
Medical Device Training Courses Medical Device HQ
Medical Devices Compliance Advanced Training Artixio
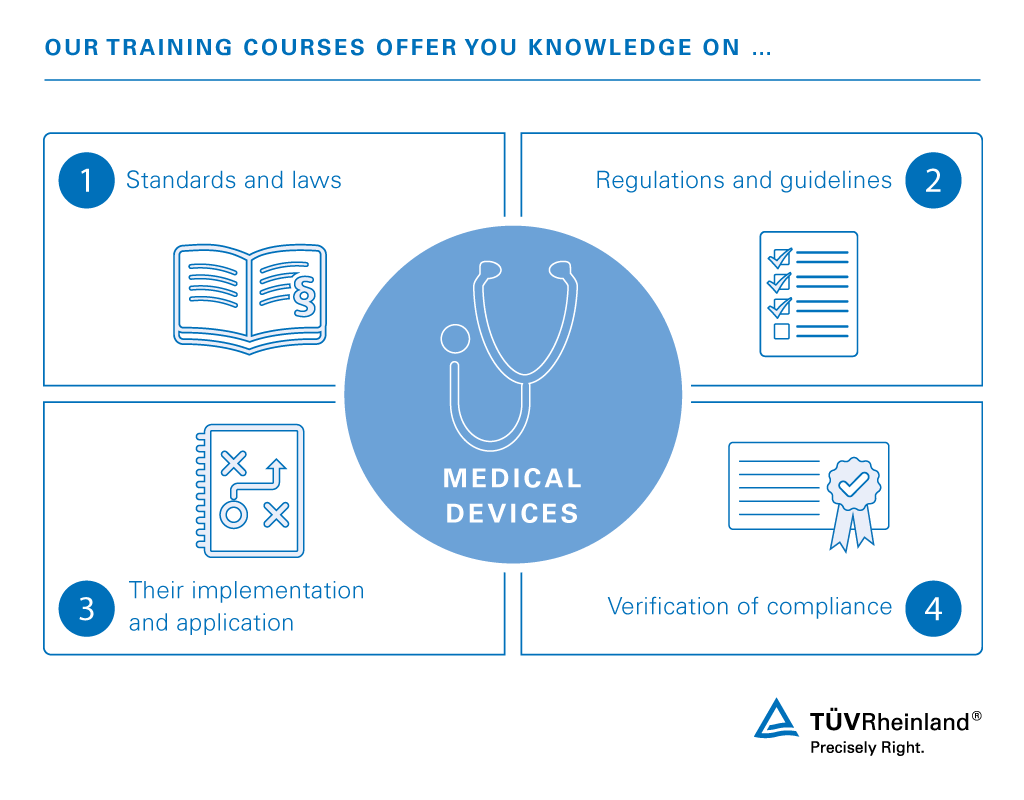
Medical Device Training TH TÜV Rheinland
Accurate Classification Of Medical Device Risk Levels Is Essential For Regulatory Oversight And Clinical Safety.
The Dual Certificate Program In Medical Devices And Pharmaceuticals.
Online Courses For Industry On Safety And Effectiveness Of Medical Devices And Exposure To Radiation From Medical Devices.
Medical Sales College Is A Licensed Private, Postsecondary Institution, Training The Next Generation Of Medical Sales Reps.
Related Post: